- Blog
- Slender the eight pages atomicgamer
- Bomb rush cyberfunk soundtrack
- Burn away tears of lifetimes fly away towards the stars
- Parasite eve ps1 cover art
- God of war ragnarok trailer
- Ublock origin for firefox
- Latin hypercube vs monte carlo
- Cyberpunk 2077 update
- I9295 zelda breath of the wild case
- Kaspersky security
- Gallon to cups
Using the spatial aspect of this method as well as the within-sample link to traditional metrics of tissue damage, we demonstrate how these data may be analyzed and used to generate new hypotheses and inform safety standards for stimulation in cortex.Īn outstanding challenge in the clinical care of cancer is moving from a one-size-fits-all approach that relies on population-level statistics towards personalized therapeutic design.
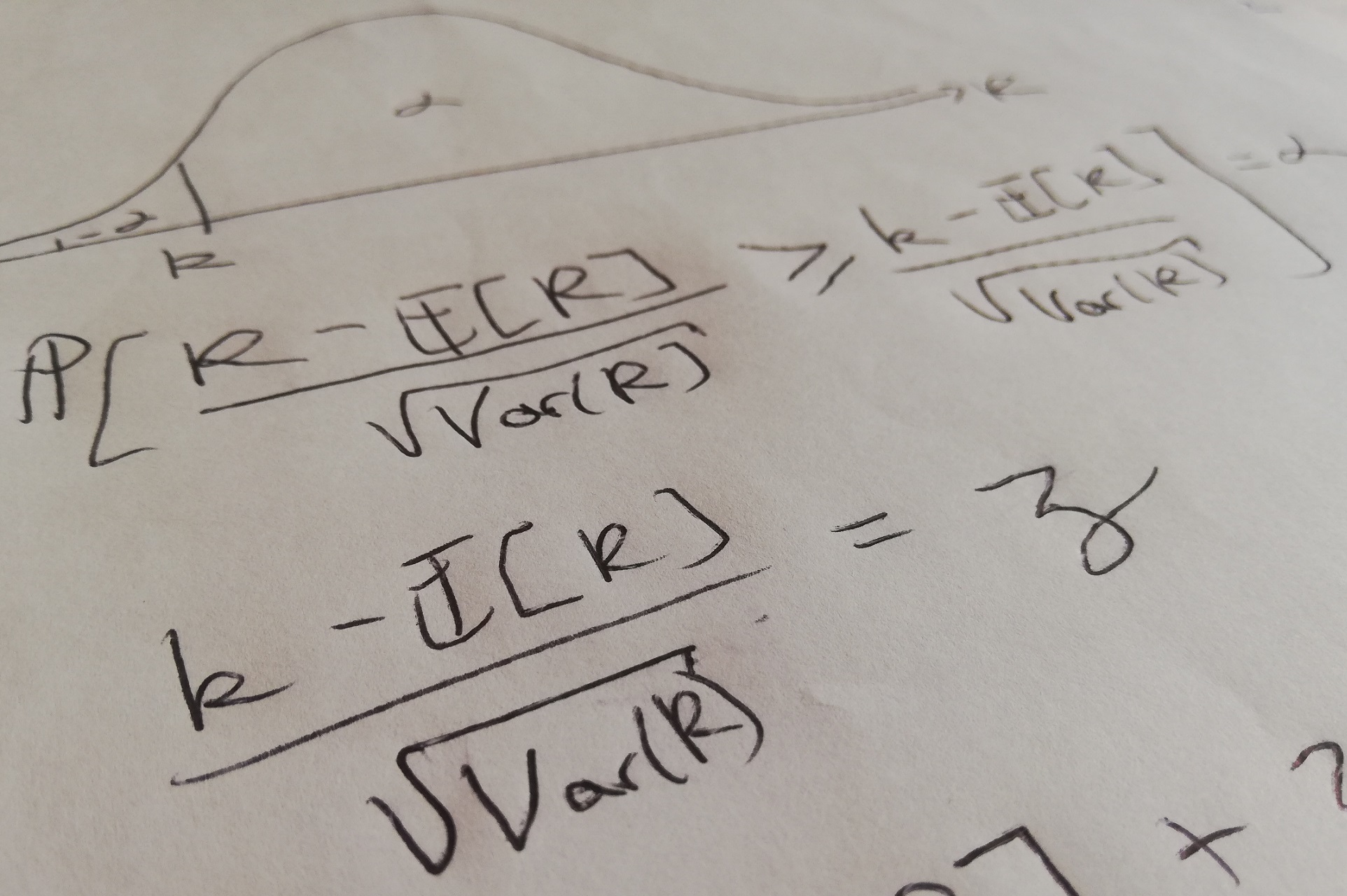
non-stimulating microwire electrode comparison and in the stimulating microwire vs. At the chronic timepoint, an increase in inflammatory and cell cycle progression related genes was observed both in the stimulating vs. At the acute timepoint, an increase in inflammatory and plasticity related genes was observed surrounding a stimulating electrode compared to a craniotomy control. Our data give a first look at unique spatial patterns of gene expression that are related to cellular processes including inflammation, cell cycle progression, and neuronal plasticity. Spatial transcriptomics was performed using Visium Spatial Gene Expression Slides (10x Genomics, Pleasanton, CA, United States), which enabled simultaneous immunohistochemistry and ST to directly compare traditional histological metrics to transcriptional profiles within each tissue sample. To explore the influence of device type and stimulation parameters, we used carbon fiber ultramicroelectrode arrays (7 μm diameter) and microwire electrode arrays (50 μm diameter) delivering charge and charge density levels selected above and below reported tissue damage thresholds (range: 2–20 nC, 0.1–1 mC/cm2). Electrical stimulation was delivered to the rat visual cortex with either acute or chronic electrode implantation procedures. Here, we demonstrate the use of spatial transcriptomics (ST) in an exploratory investigation to assess the biological response to electrical stimulation in the brain. In the three decades since the Shannon limits were reported, advances in molecular biology have allowed for more nuanced and detailed approaches to be used to expand current understanding of the physiological effects of stimulation. This set of observations and the subsequently proposed limits to safe stimulation, termed the “Shannon limits,” allow for a simple calculation (using charge per phase and charge density) to determine the intensity of electrical stimulation that can be delivered safely to brain tissue.

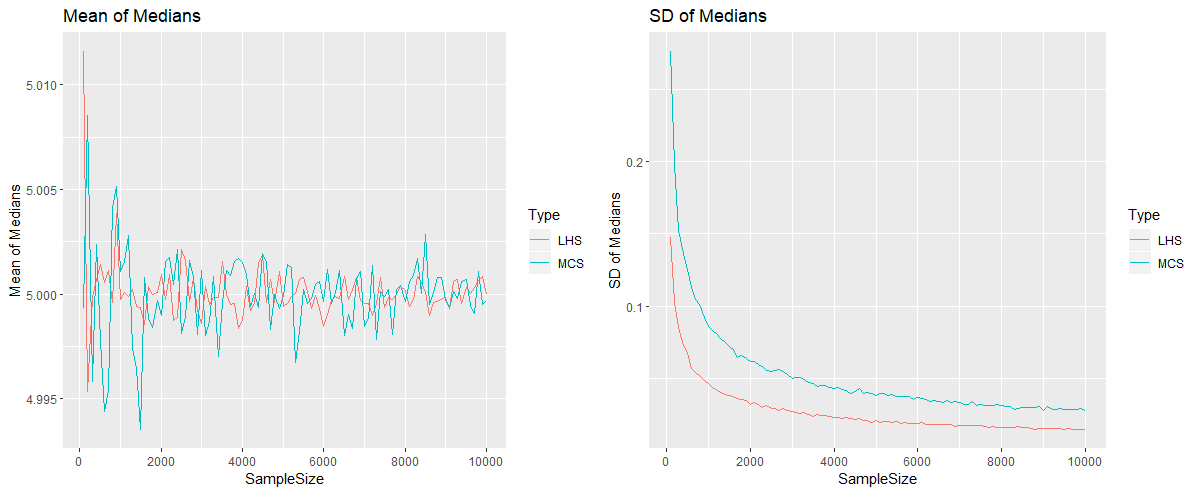
We conclude that the low discrepancy sampling and QMC technique converges substantially faster than traditional MC methods.Ĭurrent standards for safe delivery of electrical stimulation to the central nervous system are based on foundational studies which examined post-mortem tissue for histological signs of damage. We compared the number of simulations required for Monte Carlo (MC) and QMC techniques to measure the influence of input variability on the mean crack-length in an open-hole angle-ply tensile test. An Extended Finite Element Method (XFEM) method with spatially random strength fields simulates the damage initiation and evolution in a model composite. This paper demonstrates that a Quasi Monte Carlo (QMC) method, which uses a multi-dimensional low discrepancy Sobol sequence, is a computationally economical way to obtain the mean and standard deviations in cracks evolving in composites. Stochastic damage analyses require extensive sampling of random property fields and numerous replications of the underlying non-linear deterministic simulations. While the recent progressive damage analyses consider randomness in property fields, accurately quantifying the uncertainty in damage measures remains computationally expensive. Consideration of the variability in material property is critical for high-resolution simulations of damage initiation and propagation. Property variations in a structure strongly impact the macroscopic mechanical performance as regions with lower strength will be prone to damage initiation or acceleration.
- Blog
- Slender the eight pages atomicgamer
- Bomb rush cyberfunk soundtrack
- Burn away tears of lifetimes fly away towards the stars
- Parasite eve ps1 cover art
- God of war ragnarok trailer
- Ublock origin for firefox
- Latin hypercube vs monte carlo
- Cyberpunk 2077 update
- I9295 zelda breath of the wild case
- Kaspersky security
- Gallon to cups
